Types of Corrosion
Corrosion Handbook – Part 1
Corrosion damage causes costs amounting to several billion euros each year. In addition to the financial consequences, numerous dramatic damage events can be attributed to corrosion. This can significantly reduce the risk of corrosion occurring and spreading. But more about that later.
The first part of the corrosion guide gives you a general overview of corrosion and the most common types of corrosion on screw connections in the construction sector.
White rust is another type of oxidation that can form on zinc-plated components. Beyond that there is also verdigris on copper or oxide coating on aluminium. In the case of aluminium, the oxide coating acts as a barrier between air and metal and prevents further oxidation. This protects the metal from other weather influences. Many corrosion products impair the functional condition of components or systems in the environment. But not all of them are harmful.
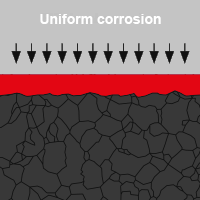
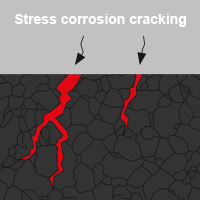
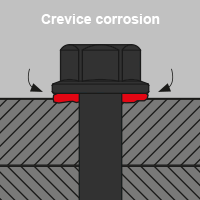
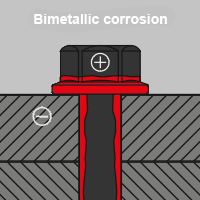
In DIN EN ISO 8044 a distinction is made between 56 types of corrosion, four of which are relevant for screw connections in the area of building envelopes and the technical building equipment:
The first part of the corrosion guide gives you a general overview of corrosion and the most common types of corrosion on screw connections in the construction sector.
What is corrosion?
DIN EN ISO 8044 describes corrosion as a “physico-chemical interaction between a metal and its environment.” According to DIN, the changed properties of the metal lead to the function of the metal, its environment or the system in which it is installed being significantly impaired.
Oxidation
The resulting corrosion products are firmly adhering, loose or soluble. For example, rust forms through a chemical reaction between iron and oxygen. This process is called oxidation: The outer surface layer of the metal absorbs oxygen. This creates an oxygen-permeable oxide layer that can chip off. Over time, the rust can eat its way into the material and break it down, weakening the affected component.
Further forms of oxidation
White rust is another type of oxidation that can form on zinc-plated components. Beyond that there is also verdigris on copper or oxide coating on aluminium. In the case of aluminium, the oxide coating acts as a barrier between air and metal and prevents further oxidation. This protects the metal from other weather influences. Many corrosion products impair the functional condition of components or systems in the environment. But not all of them are harmful.In DIN EN ISO 8044 a distinction is made between 56 types of corrosion, four of which are relevant for screw connections in the area of building envelopes and the technical building equipment:
- Uniform corrosion
- Stress corrosion cracking
- Crevice corrosion
- Bimetallic corrosion